Characterization of People Receiving 2-Drug Regimens (2DR) for HIV Management in Italy
Abstract
Objective
To describe the clinical features and real-world treatment of people living with human immunodeficiency virus (PLHIV) using fixed-dose or free combinations of 2-drug regimens (2DR) of antiretroviral therapy (ART).
Design
Italian retrospective cohort study.
Methods
Data were extracted from PLHIV who initiated or switched to 2DR: Group 1 (fixed dose), Group 2 (free combination).
Results
Group 1 was younger and more predominantly male, and had shorter time from AIDS-defining diagnosis to 2DR-ART and from diagnosis to baseline, a lower prevalence of resistance, and fewer comorbidities than Group 2. Median baseline viral load was <50 copies/mL in both groups, but Group 1 had a higher mean due to outliers.
The most common ART classes before switching to 2DR were Integrase Strand Transfer Inhibitor (INSTI)-based (48.97%), Non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI)-based (22.73%), and Protease Inhibitor (PI)-based (16.53%). Distribution varied: Group 1: INSTI-based (53.13%), NNRTI-based (24.31%), and PI-based (15.04%); Group 2: INSTI-based (29.41%), PI-based (23.53%), and NNRTI-based (15.29%).
After switching, Group 1 was on dolutegravir/lamivudine (79,33%) and dolutegravir/rilpivirine (20,67%); Group 2 mostly on INSTI-PI (52.81%), followed by NNRTI combinations, mainly with doravirine (19.10%). Duration of ART after switching was shorter in Group 1.
Conclusion
Italian PLHIV on 2DR fixed-dose combinations were younger, virologically suppressed individuals at baseline, with a shorter lead time from diagnosis, lower prevalence of resistance and lower comorbidity rate compared to those on free combinations. These findings underscore an unmet need for 2DR fixed-dose combinations, as the free combinations were predominantly utilized for more challenging populations.
Author Contributions
Academic Editor: Ian James Martins, Principal Research Fellow, Edith Cowan University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2026 Andrea Antinori, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
Andrea Antinori has received consulting fees not related to the manuscript from AstraZeneca, Bavarian Nordic, Gilead Sciences, GSK, Merck, Moderna, Pfizer, and ViiV Healthcare.
Spinello Antinori has received support for the present manuscript from MSD Italy, consulting fees from MSD, support for attending meetings from Pfizer, and participation on a DSMB from MSD.
Antonella Castagna has received consulting fees, payment for lectures or educational events, support for attending meetings, and participation in a DSMB from MSD, Gilead Sciences, Jannsen-Cilag, and ViiV Healthcare.
Benedetto M Celesia has received consulting fees, payment for lectures or presentations, and participation in a DSMB from ViiV Healthcare, Gilead Sciences, Jannsen-Cilag, MSD, and GSK Vaccines, as well as support for attending meetings from Gilead Sciences and ViiV Healthcare.
Vincenzo Esposito has received grants and consulting fees, support for attending meetings, and participation on a DSMB from Gilead, ViiV Healthcare and MSD, as well as payment for lectures, presentations and expert testimony from Gilead, ViiV Healthcare, Angelini and MSD.
Giulia C Marchetti has received payment for lectures from Gilead, ViiV Healthcare and MSD; support for attending meetings from Gilead; and for participation in a DSMB from ViiV Healthcare and Gilead.
Loredana Sarmati has received consulting fees from Gilead and Merck, as well as payment for lectures and presentations from Merck, Gilead, AbbVie, Angelini, AstraZeneca, and GSK.
Giuliano Rizzardini has received consulting fees from ViiV Healthcare and Gilead, and has participated on a DSMB for Gilead.
Anna Ancona, Stefania Grieco and Stella Grattacaso are employees at MSD and receive a salary.
Benedikt Funke BF is an employee of Merck Sharp & Dohme GmbH, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA, and may own stock and/or stock options in Merck & Co., Inc.
Maria V Cossu and Simona Di Gianbenedetto have no conflict of interest to declare.
Pier Francesco Amadeo is an employee at OPIS srl and receives a salary.
Citation:
Introduction
Three-drug antiretroviral regimens (ARV) have long been the “gold standard” first-line therapy for adult people living with human immunodeficiency virus (PLHIV) 1. However, concerns about long-term toxicities have prompted the investigation of two-drug regimens (2DR) as an alternative treatment option 2, and current EACS guidelines also recommend 2DRs 3.
Preliminary trials on early 2DR combinations for treatment-naive populations produced inconclusive outcomes due to limited sample sizes, restricted treatment options, and short durations of therapy 4. NEAT001/ANRS143, perhaps the first true trial of dual strategy, concluded that treatment of treatment-naïve PLHIV with 400 mg raltegravir (RAL) twice daily plus 800 mg darunavir and 100 mg ritonavir (DRV/r 800/100 mg) once daily as Nucleoside Reverse Transcriptase Inhibitor (NRTI)-sparing regimen was not inferior to DRV/r 800/100 mg once daily plus tenofovir disoproxil-emtricitabine (TDF/FTC) in a 245 mg and 200 mg fixed-dose combination (FDC) 5.
Lamivudine (3TC) has demonstrated favorable efficacy and safety, positioning itself as a candidate for combination therapy 6. However, early studies involved the use of 3TC with ritonavir-boosted protease inhibitors (PIs), which have been associated with metabolic adverse reactions and drug interactions. These potential drawbacks may counteract benefits expected from reduced cumulative toxicity due to decreased drug exposure 7.
Phase 3 clinical trials have yielded evidence that dolutegravir (DTG) is a promising candidate as a central component in 2DRs. The GEMINI studies support DTG/3TC as a well-tolerated option in treatment-naive PLHIV 8.
The phase III SWORD trials evaluated the efficacy and safety of once-daily dolutegravir 50 mg plus rilpivirine 25 mg (DTG/RPV) versus current ART regimen (CAR), and found DTG/RPV to be not inferior, with 95% of PLHIV in both groups achieving undetectable viremia 9.
The Non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI) doravirine (DOR) is available both as single drug for combination and as 3-drug FDC with tenofovir disoproxil fumarate (TDF) and 3TC. Evidence for its safe and effective use is available from randomized trials 10, 11, 12, and doravirine is now among the recommended drugs for first-line ART. Doravirine has little cross resistance to other NNRTI in addition to a relatively high resistance barrier 13 and can be an option for PLHIV with extensive ART history.
Sammet et al. conducted a prospective observational study of PLHIV treated with DOR + DTG as 2DR. The main reasons for choosing DOR + DTG were drug-drug interactions (DDI) (29%), tolerability (25%), and cardiovascular risk reduction (21%). DOR + DTG proved to be a durable treatment option even in extensively pretreated individuals 14.
DOR is currently under evaluation as an FDC with islatravir 15.
In Italy, a significant proportion of PLHIV is treated with 2DR, but the factors influencing this choice are still unclear 16, as well as whether the approval of 2DR FDC leads to changes regarding patient types and treatment decisions. A better understanding of patient characteristics would help to highlight residual unmet needs in modern HIV management strategies. Consequently, this study aimed to describe the baseline patient characteristics for approved FDC and free-combination dual therapies to recognize the drivers of therapeutic decisions for 2DR.
Methods
Study design and population
We conducted a retrospective medical chart review across 10 sites in Italy From 13-May-2021 to 7-July-2022. Data from 505 adult PLHIV who initiated or switched to 2DR antiretroviral therapy (ART) within the last 5 years were collected. The ART regimen had to include dual therapy based on approved fixed-dose combinations, such as DTG/RPV and DTG/3TC or any free combination dual therapy (at least one ARV drug from NNRTI, PI or Integrase Strand Transfer Inhibitor (INSTI) class of ARV drugs). No other inclusion/exclusion criteria were applied.
Eligible PLHIV were stratified in two groups: Group 1 included PLHIV who initiated or switched to approved FDC, while Group 2 included those who initiated or switched to any free-combination dual therapy.
Outcomes
The primary outcome was the baseline patient profiles in each study group. The primary analysis included demographic data, natural history of HIV acquisition, clinical presentation, drug management before and after switching/starting 2DR, and selected concomitant non-ARV medication and comorbidities.
The secondary outcome was to describe patient profiles at baseline in subgroups of PLHIV according to previous HIV medication use at index date (treatment-naïve, switching from monotherapy to 2DR, from 2DR to 2DR, from three-drug regimen (3DR) to 2DR, and from 4+DR to 2DR). The exploratory outcome was to compare the patient profiles of these subgroups. All patient profiles were analyzed except drug management before and after switching/starting 2DR.
The study also assessed the evolution/difference in the population who switched/started 2DR over time (per year and stratification group).
Variables
Variables included demographics, history of HIV acquisition, clinical presentation, drug management before initiating/switching to 2DR, comorbidities, and concomitant non-ARV medication at baseline.
History of HIV acquisition is described as age at first AIDS-defining diagnosis, time from AIDS-defining diagnosis to start/switch to 2DR, and years of known HIV status at baseline. Clinical signs focus on CD4+ count/HIV viral load (VL) at baseline. Comorbidities include cardiovascular, lipid, CNS, and liver disorders, and diabetes mellitus.
Drug management before switching/starting 2DR at index date is categorized by ART class (NRTI, NNRTI, PI, INSTI and other). Drug management after switching/starting 2DR focuses on the ART regimen at index date, and is described as the proportion of PLHIV who switched to a 2DR therapy regimen in the last 5 years and the proportion of PLHIV using each combination.
Concomitant non-ARV medications at baseline are described for PLHIV who initiated or switched to 2DR ART.
Ethics
The study protocol was reviewed by the local Health Authority. Approvals were obtained from the Ethics Committee of participating sites. Informed consent, as appropriate per local regulations, was obtained from all participants.
Statistical analysis
All statistical analyses were performed on SAS version 9.4. All variables were analyzed descriptively. Continuous data are summarized by mean, standard deviation, median, first and third quartile, minimum and maximum. Confidence Intervals (CI) (95%) are provided for each variable in groups/subgroups with more than 5 PLHIV. The exact method in case of dichotomic variables or the Goodman’s method in case of multiple categories was applied for proportions. Comparisons between stratification groups correspond to the primary analysis, and those between subgroups within stratification group to the secondary analysis.
Categorical data are presented by absolute and relative frequencies or contingency tables following the χ2 or Fisher’s exact test. Continuous data were compared using a two-sided Student’s t-test or a two-sided Wilcoxon’s rank-sum test. If the comparison involved more than two groups, ANOVA or the corresponding non-parametric Kruskal-Wallis test was used.
The evolution/difference in the population who switched /started 2DR over time based on the year of index date and stratification group is summarized. Assessment of the evolution of the 2DR populations was performed using contingency table analysis with Cochran-Armitage test for trend.
Results are based on non-missing data only (i.e., missing observations were not replaced). Participants were included in each analysis based on available assessments.
Results
Study sample
A total of 505 PLHIV charts were screened and underwent data collection. Among these, 416 received approved FDC (Group 1), while 89 received a free combination (Group 2). All were valid for analysis.
Primary analysis
The primary objective was to describe the profiles of PLHIV belonging to Group 1 and to Group 2.
Demographic overview
Overall, average age was 49.9 ± 11.96 years, and most PLHIV were male (78.22%). Group 1 was younger (49.2 ± 12.07 years vs 53.3 ± 10.87 years) and more predominantly male (81.73% vs 61.80%; p-value< 0.0001). There was no significant difference in the distribution of race and ethnicity between the two groups, with the majority of PLHIV being white (89.50%) and non-Hispanic or non-Latino (83.56%) (Table 1).
History of HIV acquisition
Overall, time from first AIDS-defining diagnosis to the start/switch to 2DR was 13.8 ± 9.16 years, and years since diagnosis of HIV acquisition 13.4 ± 9.13 at the switch to 2DR. Group 1 had a shorter time from first AIDS-defining diagnosis to the start/switch to 2DR therapy (Group 1: 13.0 ± 8.60 years; Group 2: 17.9 ± 10.55 years; p-value < 0.0001) and fewer years of HIV acquisition at baseline (Group 1: 12.5 ± 8.56 years; Group 2: 17.6 ± 10.52 years; p-value< 0.0001) (Table 1).
Table 1. Demographic overview and prior AIDS defining diagnosis| Group 1: approved fixed-dose combinations (N=416) | Group 2: free combination dual therapy (N=89) | Total (N=505) | |
| Demographic overview | |||
| Age at index date (years) | |||
| Mean (SD) | 49.2 (12.07) | 53.3 (10.87) | 49.9 (11.96) |
| Comparison of age: | |||
| p-value (Wilcoxon Test) | 0.0038 | ||
| Sex, n (%) (95% CI) | |||
| Male | 340 (81.73) (77.68; 85.33) | 55 (61.80) (50.89; 71.90) | 395 (78.22) (74.36; 81.74) |
| Female | 76 (18.27) (14.67; 22.32) | 34 (38.20) (28.10; 49.11) | 110 (21.78) (18.26; 25.64) |
| Comparison of sex: | |||
| p-value (Chi-Square Test) | <.0001 | ||
| Race, n (%) (95% CI) | |||
| Non-Hispanic or non-Latino | 343 (82.45) (77.32; 86.62) | 79 (88.76) (77.75; 94.70) | 422 (83.56) (79.04; 87.27) |
| Not reported | 28 (6.73) (4.26; 10.48) | 1 (1.12) (0.14; 8.51) | 29 (5.74) (3.66; 8.91) |
| Unknown | 5 (1.20) (0.41; 3.43) | 1 (1.12) (0.14; 8.51) | 6 (1.19) (0.45; 3.12) |
| Comparison of ethnicity: | |||
| p-value (Fisher's Exact Test) | 0.1578 | ||
| Prior AIDS defining diagnosis | |||
| Age at first AIDS-defining diagnosis (years) | |||
| Mean (SD) | 36.2 (11.00) | 35.4 (12.64) | 36.1 (11.30) |
| Comparison of age at first AIDS-defining diagnosis: | |||
| p-value (Wilcoxon Test) | 0.3421 | ||
| Time from AIDS-defining diagnosis to start/switch to 2DR (years) | |||
| Mean (SD) | 13.0 (8.60) | 17.9 (10.55) | 13.8 (9.16) |
| Comparison of time from AIDS-defining diagnosis to start/switch to 2DR: | |||
| p-value (Wilcoxon Test) | <.0001 | ||
| Years of known HIV status at baseline | |||
| Mean (SD) | 12.5 (8.56) | 17.6 (10.52) | 13.4 (9.13) |
| Comparison of years of known HIV status at baseline: | |||
| p-value (Wilcoxon Test) | <.0001 | ||
| Years of known HIV status at baseline, n (%) (95% CI) | |||
| 0-5 years | 98 (23.56) (18.64; 29.31) | 15 (16.85) (9.03; 29.28) | 113 (22.38) (17.97; 27.49) |
| 6-10 years | 101 (24.28) (19.30; 30.07) | 16 (17.98) (9.84; 30.55) | 117 (23.17) (18.70; 28.33) |
| 11-15 years | 89 (21.39) (16.69; 27.00) | 8 (8.99) (3.78; 19.89) | 97 (19.21) (15.10; 24.11) |
| 16-20 years | 55 (13.22) (9.52; 18.08) | 14 (15.73) (8.23; 27.99) | 69 (13.66) (10.20; 18.07) |
| 21 years and more | 73 (17.55) (13.26; 22.85) | 36 (40.45) (28.17; 54.06) | 109 (21.58) (17.25; 26.65) |
| Comparison of years of known HIV status at baseline: | |||
| p-value (Chi-Square Test) | <.0001 | ||
Drug management before switching/starting 2DR
Overall, the most common ART used before switching to 2DR was INSTI-based (combined with NRTIs) (48.97%), with dolutegravir/lamivudine/abacavir (DTG/3TC/ABC) as the most frequent combination (21.69%), followed by NNRTI-based (22.73%), with rilpivirine/emtricitabine/tenofovir alafenamide (RPV/FTC/TAF) as the most common (11.78%), and by PI-based 3DR (16.53%), with darunavir/cobicistat/emtricitabine/tenofovir alafenamide (DRV/c/FTC/TAF) as the most common (4.13%). Distribution varied between the two groups: Group 1 was INSTI-based (53.13%), NNRTI-based (24.31%), and PI-based (15.04%), while Group 2 was INSTI-based (29.41%), PI-based (23.53%), and NNRTI-based (15.29%).
The most frequent ARTs at baseline were DTG/3TC/ABC (25.06%), RPV/FTC/TAF (13.28%) and bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF) (9.27%) in Group 1; raltegravir (RAL) (8.24%), RAL + DRV/b (7.06%), DTG/3TC/ABC (5.88%), and RPV/FTC/TAF (4.71%) in Group 2 (Table 2).
Table 2. Prior antiretroviral therapy before switching to 2DR therapy – all medications; Enrolled population| Group 1: approved fixed-dose combinations (N=416) n (%) | Group 2: free combination dual therapy (N=89) n (%) | Total (N=505) n (%) | |
| Number of patients with at least one Prior medications before switching to 2DR therapy | 399 (95.91) (93.54; 97.60) | 85 (95.51) (88.89; 98.76) | 484 (95.84) (93.71; 97.41) |
| Prior medication category (third agent class), n (%) (95% CI) * | |||
| INSTI based | 212 (53.13) (41.51; 64.64) | 25 (29.41) (9.51; 52.87) | 237 (48.97) (38.43; 59.53) |
| NNRTI based | 97 (24.31) (14.86; 34.77) | 13 (15.29) (0.97; 35.63) | 110 (22.73) (14.32; 32.03) |
| PI based | 60 (15.04) (7.42; 24.04) | 20 (23.53) (5.58; 46.06) | 80 (16.53) (9.22; 24.93) |
| ENTRY INHIBITORS (EIs) | 1 (0.25) (0.00; 2.76) | 2 (2.35) (0.00; 14.76) | 3 (0.62) (0.00; 3.27) |
| INSTI-NNRTI | 7 (1.75) (0.00; 5.90) | 3 (3.53) (0.00; 17.18) | 10 (2.07) (0.00; 5.95) |
| INSTI-PI | 5 (1.25) (0.00; 4.97) | 16 (18.82) (2.79; 40.25) | 21 (4.34) (0.72; 9.45) |
| NNRTI-PI | 4 (1.00) (0.00; 4.48) | 0 (0.00) | 4 (0.83) (0.00; 3.70) |
| PI-EIs | 0 (0.00) | 1 (1.18) (0.00; 12.03) | 1 (0.21) (0.00; 2.28) |
| INSTI-NNRTI-PI | 0 (0.00) | 1 (1.18) (0.00; 12.03) | 1 (0.21) (0.00; 2.28) |
| NNRTI-PI--EIs | 0 (0.00) | 1 (1.18) (0.00; 12.03) | 1 (0.21) (0.00; 2.28) |
| NRTI based | 13 (3.26) (0.00; 8.40) | 3 (3.53) (0.00; 17.18) | 16 (3.31) (0.22; 7.92) |
| Prior medication category/Preferred Term * | |||
| INSTI based | |||
| BIC/FTC/TAF | 37 (9.27) (0.00; 30.77) | 2 (2.35) (0.00; 29.71) | 39 (8.06) (0.00; 26.46) |
| DTG | 1 (0.25) (0.00; 5.42) | 3 (3.53) (0.00; 35.13) | 4 (0.83) (0.00; 7.87) |
| DTG/3TC | 28 (7.02) (0.00; 26.16) | 0 (0.00) | 28 (5.79) (0.00; 21.77) |
| DTG/3TC/ABC | 100 (25.06) (0.00; 56.14) | 5 (5.88) (0.00; 44.03) | 105 (21.69) (0.00; 48.71) |
| DTG+FTC/TAF | 3 (0.75) (0.00; 8.34) | 2 (2.35) (0.00; 29.71) | 5 (1.03) (0.00; 8.73) |
| DTG+FTC/TDF | 5 (1.25) (0.00; 10.53) | 1 (1.18) (0.00; 22.97) | 6 (1.24) (0.00; 9.54) |
| EVG-c/FTC/TAF | 20 (5.01) (0.00; 21.62) | 2 (2.35) (0.00; 29.71) | 22 (4.55) (0.00; 18.96) |
| EVG-c/FTC/TDF | 2 (0.50) (0.00; 7.03) | 0 (0.00) | 2 (0.41) (0.00; 5.82) |
| RAL | 6 (1.50) (0.00; 11.50) | 7 (8.24) (0.00; 51.47) | 13 (2.69) (0.00; 14.18) |
| RAL+3TC/ABC | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| RAL+3TC/ZDV | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| RAL+FTC/TAF | 5 (1.25) (0.00; 10.53) | 1 (1.18) (0.00; 22.97) | 6 (1.24) (0.00; 9.54) |
| RAL+FTC/TDF | 1 (0.25) (0.00; 5.42) | 1 (1.18) (0.00; 22.97) | 2 (0.41) (0.00; 5.82) |
| RAL+unspecified | 2 (0.50) (0.00; 7.03) | 1 (1.18) (0.00; 22.97) | 3 (0.62) (0.00; 6.91) |
| NNRTI based | |||
| DOR/3TC/TDF | 3 (0.75) (0.00; 8.34) | 0 (0.00) | 3 (0.62) (0.00; 6.91) |
| DOR+FTC/TAF | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| EFV+3TC/ABC | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| EFV+FTC/TAF | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| EFV/FTC/TDF | 9 (2.26) (0.00; 14.08) | 3 (3.53) (0.00; 35.13) | 12 (2.48) (0.00; 13.58) |
| ETR | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| NVP | 2 (0.50) (0.00; 7.03) | 0 (0.00) | 2 (0.41) (0.00; 5.82) |
| NVP+3TC/ABC | 7 (1.75) (0.00; 12.41) | 1 (1.18) (0.00; 22.97) | 8 (1.65) (0.00; 11.00) |
| NEV+3TC+D4T | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| NEV+FTC/TAF | 4 (1.00) (0.00; 9.49) | 0 (0.00) | 4 (0.83) (0.00; 7.87) |
| NEV+FTC/TDF | 1 (0.25) (0.00; 5.42) | 1 (1.18) (0.00; 22.97) | 2 (0.41) (0.00; 5.82) |
| RPV | 2 (0.50) (0.00; 7.03) | 0 (0.00) | 2 (0.41) (0.00; 5.82) |
| RPV+3TC/ABC | 3 (0.75) (0.00; 8.34) | 0 (0.00) | 3 (0.62) (0.00; 6.91) |
| RPV/FTC/TAF | 53 (13.28) (0.00; 38.12) | 4 (4.71) (0.00; 39.82) | 57 (11.78) (0.00; 33.29) |
| RPV/FTC/TDF | 9 (2.26) (0.00; 14.08) | 2 (2.35) (0.00; 29.71) | 11 (2.27) (0.00; 12.97) |
| RPV+unspecified | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| PI based | |||
| ATVb | 12 (3.01) (0.00; 16.37) | 2 (2.35) (0.00; 29.71) | 14 (2.89) (0.00; 14.76) |
| ATVb+3TC | 4 (1.00) (0.00; 9.49) | 0 (0.00) | 4 (0.83) (0.00; 7.87) |
| ATVb+3TC/ABC | 0 (0.00) | 2 (2.35) (0.00; 29.71) | 2 (0.41) (0.00; 5.82) |
| ATZb+3TC+TDF | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| ATZb+ABC+TDF | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| ATZb+FTC/TAF | 3 (0.75) (0.00; 8.34) | 1 (1.18) (0.00; 22.97) | 4 (0.83) (0.00; 7.87) |
| DRVb | 12 (3.01) (0.00; 16.37) | 3 (3.53) (0.00; 35.13) | 15 (3.10) (0.00; 15.32) |
| DRVb+3TC | 2 (0.50) (0.00; 7.03) | 2 (2.35) (0.00; 29.71) | 4 (0.83) (0.00; 7.87) |
| DRVb+3TC/ABC | 2 (0.50) (0.00; 7.03) | 2 (2.35) (0.00; 29.71) | 4 (0.83) (0.00; 7.87) |
| DRVb+3TC/ZDV | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| DRVb/FTC/TAF | 17 (4.26) (0.00; 19.75) | 3 (3.53) (0.00; 35.13) | 20 (4.13) (0.00; 17.97) |
| DRVb+FTC/TDF | 2 (0.50) (0.00; 7.03) | 2 (2.35) (0.00; 29.71) | 4 (0.83) (0.00; 7.87) |
| DRVb+TDF | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| DRVb+unspecified | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| LPVb | 2 (0.50) (0.00; 7.03) | 1 (1.18) (0.00; 22.97) | 3 (0.62) (0.00; 6.91) |
| SQVb+3TC/ZDV | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| ENTRY INHIBITORS | |||
| MVC | 1 (0.25) (0.00; 5.42) | 2 (2.35) (0.00; 29.71) | 3 (0.62) (0.00; 6.91) |
| INSTI-NNRTI | |||
| DTG+NVP | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| DTG/RPV | 2 (0.50) (0.00; 7.03) | 1 (1.18) (0.00; 22.97) | 3 (0.62) (0.00; 6.91) |
| RAL+3TC+NVP | 2 (0.50) (0.00; 7.03) | 0 (0.00) | 2 (0.41) (0.00; 5.82) |
| RAL+3TC+RPV | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| RAL+ETR | 1 (0.25) (0.00; 5.42) | 1 (1.18) (0.00; 22.97) | 2 (0.41) (0.00; 5.82) |
| RAL+RPV | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| INSTI-PI | |||
| DRVb/FTC/TAF+DTG | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| DTG+ATVb | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| DTG/3TC/ABC+ATVb | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| DTG+ATVb | 1 (0.25) (0.00; 5.42) | 1 (1.18) (0.00; 22.97) | 2 (0.41) (0.00; 5.82) |
| DTG+DRVb | 3 (0.75) (0.00; 8.34) | 3 (3.53) (0.00; 35.13) | 6 (1.24) (0.00; 9.54) |
| RAL+DRVb | 1 (0.25) (0.00; 5.42) | 6 (7.06) (0.00; 47.89) | 7 (1.45) (0.00; 10.29) |
| RAL+FPVb | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| RAL+FTC/TDF+LPVb | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| RAL+LPVb | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| NNRTI-PI | |||
| DRVb+ETR | 4 (1.00) (0.00; 9.49) | 0 (0.00) | 4 (0.83) (0.00; 7.87) |
| PI-ENTRY INHIBITORS | |||
| DRVb+MVaC | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| INSTI-NNRTI-PI | |||
| DTG+not clear combination | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| NNRTI-PI-ENTRY INHIBITORS | |||
| DRVb/RPV/MVC | 0 (0.00) | 1 (1.18) (0.00; 22.97) | 1 (0.21) (0.00; 4.49) |
| NRTI based | |||
| 3TC | 1 (0.25) (0.00; 5.42) | 1 (1.18) (0.00; 22.97) | 2 (0.41) (0.00; 5.82) |
| ABC/3TC | 4 (1.00) (0.00; 9.49) | 1 (1.18) (0.00; 22.97) | 5 (1.03) (0.00; 8.73) |
| ABC/3TC/ZDV | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
| TAF/FTC | 2 (0.50) (0.00; 7.03) | 0 (0.00) | 2 (0.41) (0.00; 5.82) |
| TAF/FTC | 4 (1.00) (0.00; 9.49) | 1 (1.18) (0.00; 22.97) | 5 (1.03) (0.00; 8.73) |
| Missing | 1 (0.25) (0.00; 5.42) | 0 (0.00) | 1 (0.21) (0.00; 4.49) |
Drug management after switching/starting 2DR
After switching to 2DR therapy, Group 1 PLHIV were on DTG/3TC (INSTI-NRTI) (79.33%) and DTG/RPV (INSTI-NNRTI) (20.67%) combinations. Group 2 PLHIV were predominantly on INSTI-PI combination (52.81%), followed by NNRTI combinations, mainly with DOR (19.10%). The duration of the ART regimen after switching was shorter in Group 1 compared to Group 2 (9.6 ± 10.06 months vs 28.3 ± 20.30 months; p-value=0.0020) (Table 3).
Table 3. Antiretroviral therapy regimen-all medications; Enrolled Set| Group 1: approved fixed-dose combinations (N=416) n (%) | Group 2: free combination dual therapy (N=89) n (%) | Total (N=505) n (%) | |
| Patients who switched to a 2DR therapy regimen in the last 5 years | 416 (100.00) (99.12; 100.00) | 89 (100.00) (95.94; 100.00) | 505 (100.00) (99.27; 100.00) |
| Drug combination, n (%) (95% CI) | |||
| INSTI-NNRTI | 91 (21.88) (17.01; 27.66) | 13 (14.61) (7.32; 27.03) | 104 (20.59) (16.26; 25.73) |
| INSTI-NRTI | 322 (77.40) (71.57; 82.34) | 8 (8.99) (3.71; 20.22) | 330 (65.35) (59.59; 70.69) |
| INSTI-PI | 0 (0.00) | 47 (52.81) (39.16; 66.05) | 47 (9.31) (6.43; 13.29) |
| NRTI-NNRTI | 0 (0.00) | 7 (7.87) (3.05; 18.79) | 7 (1.39) (0.53; 3.56) |
| NRTI-PI | 2 (0.48) (0.09; 2.50) | 13 (14.61) (7.32; 27.03) | 15 (2.97) (1.53; 5.69) |
| PI-NNRTI | 1 (0.24) (0.03; 2.09) | 1 (1.12) (0.13; 9.21) | 2 (0.40) (0.07; 2.07) |
| Comparison of ART combination: | |||
| p-value (Fisher's Exact Test) | <.0001 | ||
| Duration of the ART regimen, months a | |||
| n | 9 | 22 | 31 |
| Mean (SD) | 9.6 (10.06) | 28.3 (20.30) | 22.9 (19.75) |
| 95% CI | 1.8; 17.3 | 19.3; 37.3 | 15.6; 30.1 |
| Median | 3.9 | 24 | 21.1 |
| Q1; Q3 | 3.7; 14.4 | 11.1; 41.7 | 3.9; 37.8 |
| Range | 0; 30 | 1; 72 | 0; 72 |
| Comparison of duration of ART regimen: | |||
| p-value (T-Test) | 0.002 | ||
| Preferred Term | |||
| Atazanavir | 0 (0.00) | 3 (3.37) (0.70; 9.54) | 3 (0.59) (0.12; 1.73) |
| Atazanavir; cobicistat | 0 (0.00) | 3 (3.37) (0.70; 9.54) | 3 (0.59) (0.12; 1.73) |
| Cobicistat; darunavir | 0 (0.00) | 41 (46.07) (35.44; 56.96) | 41 (8.12) (5.89; 10.85) |
| Darunavir | 0 (0.00) | 13 (14.61) (8.01; 23.68) | 13 (2.57) (1.38; 4.36) |
| Dolutegravir | 0 (0.00) | 55 (61.80) (50.89; 71.90) | 55 (10.89) (8.31; 13.94) |
| DTG/3TC | 330 (79.33) (75.11; 83.12) | 0 (0.00) | 330 (65.35) (61.02; 69.50) |
| DTG/RPV | 86 (20.67) (16.88; 24.89) | 0 (0.00) | 86 (17.03) (13.85; 20.60) |
| DOR | 0 (0.00) | 17 (19.10) (11.54; 28.81) | 17 (3.37) (1.97; 5.34) |
| ETR | 0 (0.00) | 1 (1.12) (0.03; 6.10) | 1 (0.20) (0.01; 1.10) |
| 3TC | 0 (0.00) | 28 (31.46) (22.03; 42.17) | 28 (5.54) (3.72; 7.91) |
| NVP | 0 (0.00) | 1 (1.12) (0.03; 6.10) | 1 (0.20) (0.01; 1.10) |
| RAL | 0 (0.00) | 13 (14.61) (8.01; 23.68) | 13 (2.57) (1.38; 4.36) |
| RPV | 0 (0.00) | 3 (3.37) (0.70; 9.54) | 3 (0.59) (0.12; 1.73) |
Clinical presentation signs
Mean HIV VL at index date was significantly different (p-value=0.0078) between the two groups, with Group 1 having a higher average than Group 2 (2161.2 ± 21272 copies/mL vs 149.8 ± 799.12 copies/mL). Nevertheless, most patients in both groups had viral load at limit of detection (19 copies/mL), as estimated by median HIV VL. The higher mean is likely due to the presence of outliers reporting higher viral loads (Figure 1).
Figure 1.Box and whisker plots for clinical presentation signs (Enrolled population)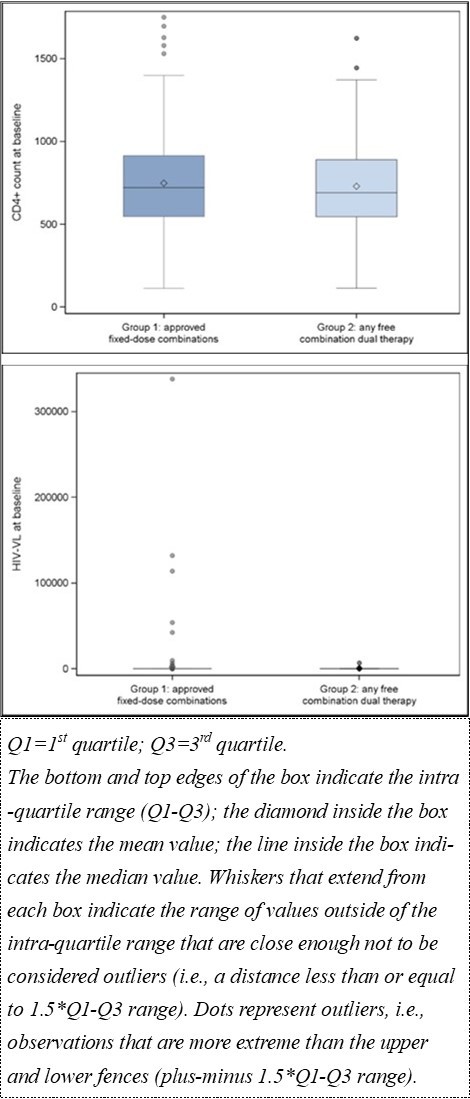
Stratifying both Group 1 and Group 2 based on their previous use of HIV drugs at index date, for Group 1, HIV-VL was relatively lower in patients who switched from 2DR to 2DR vs 3DR to 2DR (24.3 ± 39.43 copies/mL vs 26.3 ± 121.36 copies/mL). However, the comparison was not statistically significant (p-value=0.3471). For Group 2, differences in average HIV viral load were not significant (2DR to 2DR: 23.9 ± 15.44 copies/mL vs 3DR to 2DR: 295.3 ± 1164.41 copies/mL; p=0.9835).
Selected concomitant non-ARV medication
Approximately half of the enrolled population received non-ARV medication (48.51%). The most frequent in Group 1 were vitamins (18.99%), mainly cholecalciferol (17.55%), lipid modifying agents (18.75%), and agents acting on the renin-angiotensin system (17.55%). In Group 2, the most common were lipid modifying agents (32.58%), vitamins and specifically cholecalciferol (17.98%), and agents acting on the renin-angiotensin system (16.85%).
Selected comorbidities
Roughly half (54.65%) of the enrolled PLHIV presented at least one selected comorbidity. A lower proportion was reported for Group 1 (Group 1: 51.92%; Group 2: 67.42%; p-value=0.0077), and cardiovascular (24.28% and 30.3%, respectively) and lipid comorbidities (21.88% and 34.83%, respectively) were the most common in both groups.
Resistant mutation
Group 1 had a lower prevalence of resistance mutations compared to Group 2 (9.86% vs 14.61%), with the majority of cases showing PI resistance mutations in Group 1 (9.13%) and NRTI resistance mutations in Group 2 (13.48%).
Secondary analysis
The secondary objective was to describe the baseline patient profiles within subgroups according to previous HIV drug use at the index date. However, the analysis was limited to the subgroups who switched from 2DR or 3DR to 2DR, where patient numbers were sufficient to draw meaningful conclusions (Group 1: n=75 (2DR) and n=318 (3DR); Group 2: n=35 (2DR) and n=44 (3DR)).
In Group 1, PLHIV in the 3DR subgroup were younger (2DR: 53.3 ± 11.99 years; 3DR: 48.4±11.97 years; p-value=0.0013) and were more commonly male (2DR: 73.33%; 3DR: 84.28%; p-value=0.0259). In contrast, for Group 2 there was no statistically significant difference in age, sex, or ethnicity between the subgroups who switched from 2DR and 3DR.
In both stratification groups, time from AIDS-defining diagnosis to starting or switching to 2DR treatment was significantly shorter in PLHIV who transitioned from 3DR than from those who switched from 2DR (Group 1: 16.3 ± 9.23 years vs 12.4±8.09 years ; Group 2: 21.3 ± 9.90 years vs 16.0 ± 10.65 years), as well as years since known HIV status at baseline (Group 1: 15.7 ± 9.30 years vs 12.0 ± 8.05 years; Group 2: 20.9 ± 9.80 years vs 15.8 ± 10.66 years).
No statistically significant difference was found between the stratified groups for clinical presentation signs, the proportion of PLHIV who experienced any selected comorbidity, and the proportion who took any non-ARV medication.
Exploratory analysis
Comparing the two stratification groups in PLHIV switching from 2DR and 3DR to 2DR, males were more represented in Group 1 (2DR: 73.33%; 3DR: 268 84.28%) than in Group 2 (2DR: 54.29%; 3DR: 70.45%). Additionally, in both subgroups, time from AIDS-defining diagnosis to starting or switching to 2DR and the number of years with known HIV status at baseline were significantly lower in Group 1 time since diagnosis: 16.3 ± 9.23 years (2DR); 12.4 ± 8.09 years (3DR); years with known HIV status: 15.7 ± 9.30 years (2DR); 12.0 ± 8.05 years (3DR) compared to Group 2 (time since AIDS-defining diagnosis: 21.3 ± 9.90 years (2DR); 16.0 ± 10.65 years (3DR); years with known HIV status: 20.9 ± 9.80 years (2DR); 15.8 ± 10.66 years (3DR)).
At baseline, CD4+ count was not significantly different between stratification groups for PLHIV switching from 2DR to 2DR (p-value=0.6178) and from 3DR to 2DR (p-value=0.8688). HIV viral load at baseline was significantly different between stratification groups only in PLHIV switching from 3DR to 2DR (Group 1: 26.3 ± 121.36 copies/mL; Group 2: 295.3 ± 1164.41 copies/mL; p-value=0.0403). Also, the proportion of PLHIV who took any non-ARV medication was not significantly different between subgroups. The proportion of PLHIV who experienced any selected comorbidity was significantly higher in Group 2 for PLHIV switching from 2DR to 2DR (Group 1: 54.67%; Group 2: 74.29%; p-value = 0.0495).
Finally, an explorative analysis of the evolution of the 2DR population showed an increasing trend in the number of PLHIV who switched/started 2DR over time in both groups.
Discussion
This study highlights the significance of considering the baseline characteristics of PLHIV while developing HIV management strategies. To the best of our knowledge, this is the first real-world study conducted in Italy to assess the baseline profiles of PLHIV who are on 2DR FDC compared to those on 2DR free-combination therapies. Statistically significant differences were observed between the two groups with respect to age, gender, time from AIDS-defining diagnosis to start/switch to 2DR, years of known HIV status at baseline, HIV VL, ART regimen and selected comorbidities.
Many PLHIV (n=416) initiated FDC therapy (DTG/RPV or DTG/3TC), and these participants were more likely white males. This is in line with previous evidence that reported the majority of people with HIV treated with FDC DTG/3TC being male and white 17. However, as per the report of Centre for Disease Control and Prevention (CDC), Black/African American and Hispanic/Latino communities are disproportionately affected by HIV compared to other racial/ethnic groups 18. Group 1 PLHIV were older, with an average age of 49.2±12.07 years compared to 32.5 years in the GEMINI 1 and 2 clinical trials conducted on treatment-naïve PLHIV 19, but of similar age compared to other real-world data; 48.6 years (EUROSIDA HIV) 20 and 51 years 21.
Group 1 had significantly shorter time from AIDS-defining diagnosis to start of 2DR and fewer years of known HIV status than those in Group 2. In Group 1, the median time from AIDS-defining diagnosis to start of 2DR was 11.5 years, compared to 14 years in the LAMIDOL study 22 and 15 years in the DOLAMA study 23.
One of the significant factors that influences the HIV therapeutic choice is HIV VL at treatment initiation. In our cohort, 399 PLHIV had a median HIV VL<50 copies/mL at baseline. There was a significant difference in mean baseline HIV VL between the two groups, with Group 1 having a higher average than Group 2. From the results, it can be interpreted that the PLHIV were under viral suppression and presented a lower risk of disease progression, irrespective of the combination therapies. The efficacy and safety of DTG plus 3TC for treating PLHIV without resistance mutations were settled by the TANGO randomized trial 24. These findings support the use of 2DR as a switch option for PLHIV with viral suppression on a 3- or 4-DR who wish to receive fewer ARTs due to treatment complexity, avoidance of potential drug-associated toxicity, risk of drug–drug interactions, or cost. Clinicians should consider viral loads at baseline for tailoring HIV management strategies to ensure optimal viral suppression and prevent the emergence of drug-resistant strains. Of note, the greater prevalence of resistant mutations in Group 2 suggests that clinical needs have not yet been fully addressed with approved 2DR FDC, and warrants investigation of potential further 2DR strategies such as DOR/ISL, which has demonstrated high potency even in the presence of drug-resistance mutations 25.
PLHIV in both cohorts were medically complex, with diagnoses of hypertension, hypercholesterolemia, dyslipidemia and vitamin D deficiency. Another study also indicated that multi-comorbidities were higher in PLHIV who switch from 2DR compared with 3DR 26.
The average duration of the ART regimen after switching differed significantly between the groups, with shorter duration in Group 1. According to the ICONA report, gender, type of treatment and time spent on HIV management were independent factors associated with discontinuation of ART regimens 27. However, our current study did not explore the reason behind switching ART.
PLHIV who switched from 2DR were found to be significantly older, more experienced at the index date, and less likely to be male when compared to individuals on a 3DR. These findings are consistent with those reported by Ruzika et al 26.
Our study highlights that individuals who switch to 2DR may be difficult to treat, particularly if they were on free combination therapies due to the presence of resistant mutations. The need for more effective and tailored treatment options is increasingly recognized, particularly for individuals who may not respond optimally to standard triple therapy. Currently approved FDC may not adequately address the clinical complexities of various patient populations, thereby reinforcing the unmet need for more innovative therapeutic options. Ongoing clinical development of novel 2DR seeks to fill this gap. The combination of doravirine and islatravir (DOR/ISL) and bictegravir and lenacapavir (BIC/LEN) administered once daily are under investigation for their potential to provide robust virologic suppression with a favorable tolerability profile 28, 29. Furthermore, the weekly dosing option of ISL in combination with LEN is being explored to enhance patient adherence while maintaining viral suppression 30. These advancements in 2-drug regimens will address the significant unmet needs associated with current treatment paradigms, reducing treatment burden and thereby advancing the overall management of HIV.
Although our study's retrospective data are not fully representative of the general Italian PLHIV population, it aimed to describe the baseline characteristics of PLHIV on 2DR FDC and free combination therapies, with DOR as the last NNRTI launched on the market at the time of enrollment. We assume that a similar relationship between the two comparison groups would hold in the general population as well.
This medical chart review renders the study results representative of real-world clinical practice. The study was performed in Italy, suggesting that the results may be more representative of European countries and not of the broader population and are specific to the stratification groups mentioned. The enrolment of a low percentage of women under both combination therapies suggests that the study demographics do not fully represent the Italian population of PLHIV. Data on patient adherence and virological failure with previous HIV drug use at baseline and reasons for switching to 2DR were not collected at baseline, hence the study was unable to evaluate treatment patterns based on the aforementioned variables. The majority of PLHIV had a HIV-VL<50 copies/mL, which did not allow any baseline consideration for PLHIV with very high viral loads.
Conclusion
This study was the first step in understanding the baseline profiles and medical needs of PLHIV who started 2DR in Italy. It provides a foundation for future studies to further analyze reasons for switching therapy, as well as to identify those PLHIV that may most benefit from current and future 2DR therapies. In conclusion, PLHIV in Italy on currently approved FDC 2DR therapies were younger, virologically suppressed individuals at baseline with shorter time from prior AIDS-defining diagnosis, lower prevalence of resistance, and lower comorbidities rates compared to individuals on free dual combination therapies. This highlights existing unmet needs of PLHIV with currently approved FDC, indicating a pressing need for ongoing research and the development of tailored treatment strategies that cater to the diverse clinical profiles of this population.
Ethics
The study protocol was reviewed by the local Health Authority. Approvals were obtained from the Ethics Committee of participating sites. Informed consent, as appropriate per local regulations, was obtained from all participants. Data is available upon request.
All authors conceptualized, critically revised the manuscript for important intellectual content, and approved the final manuscript.
Source of Funding
Funding for this research was provided by MSD Italia Srl.
Acknowledgments
Medical writing and statistical analyses were provided by OPIS srl. This assistance was funded by MSD Italia.
References
- 1. (2018) US Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV.
- 2.Pau A K, George J M. (2014) Antiretroviral therapy: current drugs.Infect Dis Clin North Am. 28(3), 371-402.
- 4.Baril J G, Angel J B, Gill M J, Gathe J, Cahn P et al. (2016) Dual Therapy Treatment Strategies for the Management of Patients Infected with HIV: A Systematic Review of Current Evidence in ARV-Naive or ARV-Experienced, Virologically Suppressed Patients.PLoSOne. 11-2.
- 5.Raffi F, Babiker A G, Richert L, Molina J M, George E C et al. (2014) NEAT001/ANRS143 Study Group. Ritonavir-boosted darunavir combined with raltegravir or tenofovir-emtricitabine in antiretroviral-naive adults infected with HIV-1: 96 week results from the NEAT001/ANRS143 randomised non-inferiority trial.Lancet. 384(9958), 1942-51.
- 6.Cahn P, Andrade-Villanueva J, Arribas J R, Gatell J M, Lama J R et al. (2014) Dual therapy with lopinavir and ritonavir plus lamivudine versus triple therapy with lopinavir and ritonavir plus two nucleoside reverse transcriptase inhibitors in antiretroviral-therapy-naive adults with HIV-1 infection: 48 week results of the randomised, open label, non-inferiority GARDEL trial.Lancet Infect Dis. 14(7), 572-80.
- 7.Lv Z, Chu Y, Wang Y. (2015) HIV protease inhibitors: a review of molecular selectivity and toxicity.HIV AIDS(Auckl). 7, 95-104.
- 8.Cahn P, Madero J S, Arribas J R, Antinori A, Ortiz R et al. (2020) . Durable Efficacy of Dolutegravir Plus Lamivudine in Antiretroviral Treatment-Naive Adults With HIV-1 Infection: 96-Week Results From the GEMINI-1 and GEMINI-2 Randomized Clinical Trials.JAcquirImmuneDeficSyndr 83(3), 310-8.
- 9.Aboud M, Orkin C, Podzamczer D, Bogner J R, Baker D et al. (2019) Efficacy and safety of dolutegravir-rilpivirine for maintenance of virological suppression in adults with HIV-1: 100-week data from the randomised, open-label, phase 3 SWORD-1 and SWORD-2 studies.Lancet HIV.
- 10.Molina J M, Squires K, Sax P E, Cahn P, Lombaard J et al. (2020) Doravirine versus ritonavir-boosted darunavir in antiretroviral-naive adults with HIV-1 (DRIVE-FORWARD): 96-week results of a randomised, double-blind, non-inferiority, phase 3 trial.Lancet HIV.
- 11.Johnson M, Kumar P, Molina J M, Rizzardini G, Cahn P et al. (2019) Switching to doravirine/lamivudine/tenofovir disoproxil fumarate (DOR/3TC/TDF) maintains HIV-1 virologic suppression through 48 weeks: results of the DRIVE-SHIFT trial.JAcquirImmuneDeficSyndr. 81, 463-472.
- 12.Orkin C, Squires K E, Molina J M, Sax P E, Wong W W et al. (2019) Doravirine/lamivudine/tenofovir disoproxil fumarate is non-inferior to efavirenz/emtricitabine/tenofovir disoproxil fumarate in treatment-naive adults with human immunodeficiency virus-1 infection: week 48 results of the DRIVE-AHEAD trial.Clin Infect Dis. 68, 535-544.
- 13.Sterrantino G, Borghi V, Callegaro A P, Bruzzone B, Saladini F et al. (2019) Prevalence of predicted resistance to doravirine in HIV-1-positive patients after exposure to non-nucleoside reverse transcriptase inhibitors.Int JAntimicrobAgents. 53, 515-519.
- 14.Sammet S, Touzeau-Römer V, Wolf E, Schenk-Westkamp P, Romano B et al. (2023) The DoDo experience: an alternative antiretroviral 2-drug regimen of doravirine and dolutegravir.Infection. 51(6), 1823-1829.
- 15.Mills A M, Rizzardini G, Rampogal M N, Osiyemi O O, Bogner J R et al.Switch to fixed-dose doravirine (100 mg) with islatravir (0·75 mg) once daily in virologically suppressed adults with HIV-1 on bictegravir, emtricitabine, and tenofovir alafenamide: 48-week results of a phase 3, randomised, controlled, double-blind, non-inferiority trial.Lancet HIV.Volume 11. , Issue 6, 357-368.
- 16.Gianotti N, Lorenzini P, Cozzi-Lepri A, A De Luca, Madeddu G et al. (2019) Durability of different initial regimens in HIV-infected patients starting antiretroviral therapy with. CD4+ counts <200 cells/mm3 and HIV-RNA >5 log10 copies/mL.JAntimicrobChemother 74(9), 2732-41.
- 17.Patel R, Evitt L, Mariolis I, S Di Giambenedetto, d’Arminio Monforte A et al. (2021) HIV Treatment with the Two-Drug Regimen Dolutegravir Plus Lamivudine in Real-world Clinical Practice: A Systematic Literature Review.Infect Dis Ther. 10(4), 2051-70.
- 19.Cahn P, Madero J S, Arribas J R, Antinori A, Ortiz R et al. (2019) Dolutegravir plus lamivudine versus dolutegravir plus tenofovir disoproxil fumarate and emtricitabine in antiretroviral-naive adults with HIV-1 infection (GEMINI-1 and GEMINI-2): week 48 results from two multicentre, double-blind, randomised, non-inferiority, phase 3 trials.Lancet. 393(10167), 143-55.
- 20.Pelchen-Matthews A, Ryom L, ÁH Borges, Edwards S, Duvivier C et al. (2018) Aging and the evolution of comorbidities among HIV-positive individuals in a European cohort.AIDS. 32(16), 2405-16.
- 21.Borghetti A, Baldin G, Lombardi F, Ciccullo A, Capetti A et al. (2018) Efficacy and tolerability of lamivudine plus dolutegravir as a switch strategy in a multicentre cohort of patients with suppressed HIV-1 replication.HIV Med.
- 22.Baldin G, Ciccullo A, Borghetti A, S Di Giambenedetto. (2019) Virological efficacy of dual therapy with lamivudine and dolutegravir in HIV-1-infected virologically suppressed patients: long-term data from clinical practice.J.Antimicrob.Chemother. 74(5), 1461-3.
- 23.Hidalgo-Tenorio C, Cortés L L, Gutiérrez A, Santos J, Omar M et al. (2019) DOLAMA study: Effectiveness, safety and pharmacoeconomic analysis of dual therapy with dolutegravir and lamivudine in virologically suppressed HIV-1 patients.Medicine(Baltimore).
- 24.Scott L J. (2020) Dolutegravir/Lamivudine Single-Tablet Regimen: A Review in HIV-1 Infection.Drugs. 80(1), 61-72.
- 25.Lai M T, Feng M, Xu M, Ngo W, Diamond T L et al. (2022) Doravirine and Islatravir Have Complementary Resistance Profiles and Create a Combination with a High Barrier to Resistance.AntimicrobAgentsChemother.
- 26.Ruzicka D J, Kamakura M, Kuroishi N, Oshima N, Yamatani M et al. (2022) Characteristics of 2-drug regimen users living with HIV-1 in a real-world setting: A large-scale medical claim database analysis in Japan.PLoSOne. 17-6.
- 27.d’Arminio Monforte A, Lepri A C, Rezza G, Pezzotti P, Antinori A et al. (2000) Insights into the reasons for discontinuation of the first highly active antiretroviral therapy (HAART) regimen in a cohort of antiretroviral naïve patients. I.CO.N.A. Study Group. Italian Cohort of Antiretroviral-Naïve Patients.AIDS. 14(5), 499-507.
- 28.Molina J M, Rizzardini G, Orrell C, Afani A, Calmy A et al. (2024) Switch to fixed-dose doravirine (100 mg) with islatravir (0·75 mg) once daily in virologically suppressed adults with HIV-1 on antiretroviral therapy: 48-week results of a phase 3, randomised, open-label, non-inferiority trial.The LancetHIV. 11, 369-379.
